Fajan’s rule-definition-postulates and examples
Fajan’s rule definition in chemistry
It is a common experience that many times an ionic bond has some covalent character and a covalent bond has some ionic character.
Fajan’s rule
is related to the covalent character in ionic bond or ionic compounds.
It has been experimentally observed that a polar covalent bond is more stable than a pure covalent or a pure ionic bond .
It has been experimentally observed that a polar covalent bond is more stable than a pure covalent or a pure ionic bond .
The higher
the degree of ionic polarization , the greater is the stability of the polar
covalent bond .
The extent
of polarization depends , evidently, on the polarizing power of the cation and
the polarizability of the anion.
The rules
regarding to the polarization, is known
as Fajan’s rule .
Fajan’s rule postulates with example in chemistry
Fajan gave some postulates or condition to explain how does covalent character gradually increase in ionic bond or ionic compounds . The main postulates of Fajan’s rule are discussed below.
Postulate-I
: Size of cation .
The cation
with smaller size have higher polarizing power, that is , they cause
polarization of electron charge cloud of an anion to a greater extent.
Hence, with
decreasing cationic size, the covalent character of the concerned compounds for
a particular anion increases.
For example, the cationic size [ M 2+ ]of group-II alkaline metal elements gradually increases from top to bottom along the group . Hence covalent character decreases from top to bottom.
Hence,
covalent character of group-II metal halides decreases from top to bottom along the group.
Postulate-II: Size of anion .
The anions
with large size have high polarizability . That is, their electron charge cloud
can be deformed by cation easily.
Hence, with
increasing anionic size , the covalent character of the related compound
increases.
For example,
the size of halide ions gradually increase from top to bottom and hence the
covalent character of halogen compounds for a particular cation are
increased from top to bottom .
For example, covalent character of different
calcium halides increases from F –
to I –.
Postulate-III:
Charge of cation or anion or both.
The degree of polarization of anion by
cation enhance with enhancing the extent of charge on cation or anion or both.
Because, the
electrostatic forces which cause the polarization, would increase with increase
in the charge on the ions .
Therefore,
with increase in charge on ions, the covalent character of the producing
compounds increases.
For example, AlCl3 is more covalent than MgCl2
For example, AlCl3 is more covalent than MgCl2
Postulate-IV
: The number of electrons in the outer most inert shell of cation.
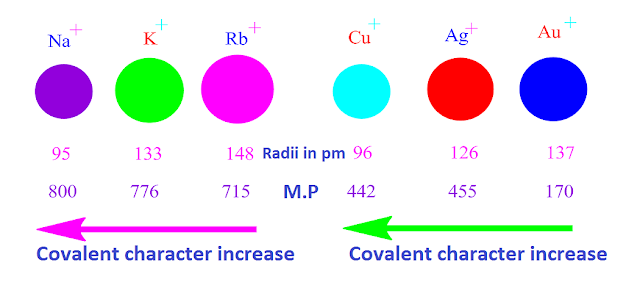
It has been found that, cations with pseudo inert gas
configuration, such as, ns2 p6 d 10, or with inert pair configuration, such
as, (n-1)d 10 ns 2, have
high polarizing power ,while cations with noble gas configuration, such as, ns2 np6, have low polarizing power.
That is due
to greater effective nuclear charge [ low shielding effect of f-orbital ] in
the former cases and smaller effective nuclear charge in the later cases.
For the
above reason, chloride compounds of Cu,
Ag and Au [with electronic configuration,
such as, ns2 p6 d 10] are more covalent
than chloride compounds of Na , K and Rb [ with electronic configuration ,ns2
np6 ] respectively.
Postulate-V
: Dielectric constant of medium.
The value of
dielectric constant of polar medium is high . In polar medium, due to high
dielectric constant, polarization does not occur .
But in non-polar medium, due
to lower dielectric constant, polarization occurs easily.
That is, in
non-polar medium, the covalent character of a compound increases.
Examples of Fajan’s rule
Why does MgCl2 is more covalent than NaCl?
The degree of polarization of anion by
cation enhance with enhancing the extent of charge on cation or anion or both.
Because, the
electrostatic forces which cause the
polarization, would increase with increase in the charge on the ions .
Therefore,
with increase in charge on ions, the covalent character of the producing compounds increases.
Since, the
extent of charge on Mg 2+ cation is higher than Na + cation , so MgCl2 is more covalent than NaCl.
Why does AgI is more covalent than AgCl ?
The anions
with large size have high polarizability. That is, their electron charge cloud
can be deformed by cation easily.
Hence, with
increasing anionic size, the covalent character of the related compound
increases.
For example,
the size of halide ions gradually
increase from top to bottom and hence the covalent
character of halogen compounds for a particular cation are increased
from top to bottom.
Consequently,
AgI is more covalent than AgCl .
Summary
Fajan’s rule definition in chemistry .
Fajan’s rule postulates with example in chemistry .
Examples of Fajan’s rule
Why does AgI is more covalent than AgCl ?
Why does MgCl2 is more covalent than NaCl ?













No comments